Chinese Journal of Tissue Engineering Research ›› 2015, Vol. 19 ›› Issue (8): 1277-1283.doi: 10.3969/j.issn.2095-4344.2015.08.024
Previous Articles Next Articles
Phase III study on surface construction and biocompatibility of polymer materials as cardiovascular devices: coagulant and anti-coagulant surface modification
Chen Bao-lin1, Wang Dong-an2, 3
- 1 Bureau of Scientific Research, Hulunbuir College, Hulunbuir 021008, Inner Mongolia Autonomous Region, China; 2 Institute of Polymer Science, Zhejiang University, Hangzhou 310027, Zhejiang Province, China; 3 Department of Pharmaceutical Sciences, University of Tennessee Health Science Center, Memphis, Tennessee 38163, USA
-
Revised:2014-12-30Online:2015-02-19Published:2015-02-19 -
About author:Chen Bao-lin, Professor, Bureau of Scientific Research, Hulunbuir College, Hulunbuir 021008, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Chen Bao-lin, Wang Dong-an. Phase III study on surface construction and biocompatibility of polymer materials as cardiovascular devices: coagulant and anti-coagulant surface modification[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1277-1283.
share this article

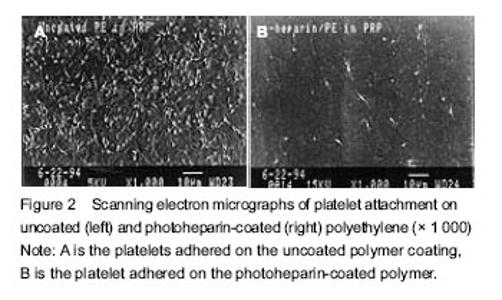
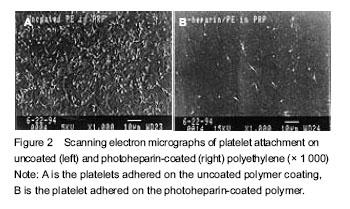
The interaction between the implant and the blood All synthetic implants (including so-called "inert material") are present as endogenous substances in the contact with the host blood environment, and may cause different levels of host response, such as deposition of plasma proteins and platelets, infiltration of neutrophils and monocytes, as well as migration of endothelial cells and smooth muscle cells. In fact these host responses have very complex mechanisms, which play a crucial role on the in vivo implantation[2]. Protein adsorption When the implants are in contact with the blood, plasma proteins are immediately deposited on the surface of the implants. A large amount of serum proteins such as albumin, fibrinogen and immunoglobulin G may adsorb to the material surface. According to the Vroman effect, these proteins are redistributed due to different biochemical affinities and charge affinities on the material surface. Both platelets and blood cells are binding with the proteins, rather than the implants directly, so the concentration of the ligand expressed on the protein surface plays a crucial role on the viability of the inhibitors. Fibrin, fibronectin, laminin and vitronectin contain Arg-Gly-Asp (RGD) sequence region, which binds with the GIIb/IIIa receptor complex on the platelet surface[3]. Therefore, the activation of platelet deposition may be affected by protein adsorption that occur previously. Other plasma proteins including complement components may also be activated on the implant surface. Platelet deposition Early platelet deposition is the result of mutual interactions between the surface protein receptors and the proteins adhered to the implant surface, it also includes pure physical absorption. The formation of platelet/protein complexes is mediated by platelet membrane glycoprotein of von Wilkbrand factor. Platelet conformations begin to change after deposition and release a series of biologically active substances, including serotonin, adrenocorticotropic hormone, ADP and thromboxane A2. These substances can activate other platelets and increase thrombin generation[4]. Infiltration of neutrophils and monocytes Acute inflammation is caused by the neutrophils adsorbed onto the surface of the artificial implants, which is mediated by C5a, Leukotrienc B4 and other strong chemoattractants. Neutrophils can be adsorbed to the agglutinators that are encapsulated in and out of implants, and also adhere to the endothelial cells around the anastomosis stoma through the strong adhesion of the leukocyte-endothelial cell adhesion molecule. The activated neutrophils release oxygen free radicals and protease, which can cause the substrate degradation and protein vascular graft from complete endothelialization. Furthermore, the monocytes in blood circulation are prone to adsorb to wounds or endothelium regeneration site. There are many kinds of activating factors in plasma, due to these factors, monocytes differentiate into macrophages and participate in the host chronic inflammatory response. Macrophages release oxygen free radicals and proteases, which can function in the tissue/implant/blood interface for a long term, some materials are even phagocytized by macrophages. At the same time, macrophages release mitogens to stimulate the proliferation of smooth muscle cells, endothelial cells and fibroblasts at quiescent stage[5-19]. Ingrowth of endothelial cells and smooth muscle cells Autologous vascular endothelial cells are arranged orderly and secrete bioactive substances to prevent thrombosis, meanwhile initialize fibrin dissolution, prevent the proliferation of smooth muscle cells and maintain normal blood flow. However, artificial implants often appeared endothelial ingrowth, in which the depth does not exceed 1.0-2.0 cm over the anastomosis stoma. There are some damaged or unfused endothelial cells around the anastomosis stoma, but the biochemical properties change and are like to induce thrombus and promote the growth of smooth muscle. The proliferation and thickening of subintimal smooth muscle cells are more evident at the anastomosis stoma, where chronic cell retrograde and chronic inflammation occur. Smooth muscle cells, inflammatory mononuclear phagocytes and foreign macrophages also produce a series of growth regulating substances (factors), which continuously stimulate the proliferation of smooth muscle cells and produce extracellular matrix components. The hyperplasia of smooth muscle cells is usually considered as the leasing cause of endometrial hypertrophy[3]. Anti-coagulant surface modification of biomaterials As mentioned above, when the untreated synthetic materials are in contact with the blood, they produce a series of biochemical reactions, which certainly cause serious harm on the host, even life-threatening[20]. To solve this situation, patients need anticoagulant drugs such as heparin during the interventional treatment or artificial graft implantation. However, the administration induces very serious adverse reactions, even fatal consequences. Therefore, improving anticoagulant properties of the implants is a radical approach. Changing the nature of the materials can improve blood compatibility and inevitably affect their physical-mechanical properties and chemical properties. Therefore, surface modification method that fully retains the material properties has been widely used. Among the surface modification methods, purely physical modification is the dominant due to simple operation, low cost, and less harmful residue. The schematic diagram of surface modification to improve blood compatibility of the catheters is shown in Figure 1. Anti-coagulant surface coatings can be divided into short-term, long-term and permanent according to the duration of exposure to blood. Anti-adhesion coating Anti-adhesion coating is a kind of short-term coating, which is mainly used to exclude the adhesion of platelets, proteins and other coagulation substances[21]. Due to the short-term use, this coating requires no heparin and other bioactive substances. Such short-term anti-coagulant coating materials have been introduced into commercial production, such as Slip-Coat polymer coating (STS Biopolymers Company, Henrietta, NY, USA), light grafting hydrogel surface system (SurModics Company, Eden Prairie, MN, USA), SPI-Polymer (Spire Corp Company, Bedford, MA, USA), and Hydromer (Hydromer Company, Branchburg, NJ, USA). These products are consisted of surface-anticoagulant coronary catheter, guiding catheter, angioplasty catheter, balloon dilator, guider, as well as topical intubation. As the interventional catheters, the surface smoothness[22-28] is also essential in addition to anticoagulant surface modification. A previous study of Target Therapeutics (Fremont, CA, USA) has shown that, improving surface smoothness of the catheter can not only reduce the damage to surrounding tissue in the catheter insertion process, but also reduce surface adhesion of blood tangible materials. The photosensitive hydrogel coating has an antiplatelet adhesion effect and even exceeds the surface modification through the heparin fixation (without special smoothing process)[4]. Bioactive coating Bioactive coating contains heparin and other bioactive factors, and allows a long-term utilization of interventional therapy. Generally speaking, bioactive coating has a better anticoagulant effect than simple anti-adhesion surface modification, and the beneficial effect is more apparent for the interventional therapy. The heparin-contained surface coating avoids the presence of microthromb shedding and the blockage of cardiovascular vessels caused by conventional coating. The heparinized coating-modified medical devices include coronary bypass oxygenator, central venous catheters, coronary stents and hemolytic balancing device[4]. Heparin itself is a highly charged molecule and can disrupt the interaction between thrombin and factor Xa in the “waterfall model” generated by coagulant fibrinogen[21, 29-31], so the heparinized coating prevents platelet adhesion and activation (Figure 2). Heparinized coating is shown to inhibit overall degree of coagulation and prolong acute coagulant time in clinical experiments[4, 32]. Heparinized coating materials have been commercially produced, such as Carmeda BioActive Surface (CBAS; Carmeda AB Company, Stocknolm), surgical Duraflo surface systems (Baxter International Company, Deerfield, IL, USA), PhotoLink coating (SurModics) and Medi-Coat hydrogel surface system (STS). Surface coating promoting the growth of host cells The surface coating modification of permanent implantable medical devices (such as vascular grafts, vascular stents and heart valves sutural margin) requires anti-coagulation functions and strong anti-immune recognition functions. The most direct way to hinder the immune system in human body is to modify and form “novel” material surface in contact with the human body, using the autologous tissue cells. In brief, human endothelial cells are introduced to the inner wall of artificial blood vessel through a tissue engineering method, to impede anticoagulant and immune response; in order to stimulate the proliferation and migration of endothelial cells and prevent excessive proliferation of smooth muscle cells, it is feasible to stimulate excessive secretion of growth factors and growth inhibiting factors from endothelial cells, by transgenic technology[33-39]. Such engineered blood vessel is different from human blood vessel, because the engineered vessels need prosthesis support to maintain its structure and do not contain elastin[40-42]. The surface morphology of biomaterials by endothelial tissue engineered modification (including uncoated graft) is shown in Figure 3[43-48]. Other blood compatible coatings Other blood compatible coatings include the phosphorylcholine-contained surface coating, albumin surface coating, and the plasminogen activator-contained surface coating. Wherein phosphorylcholine itself is present as an alternative to the outer membrane of a cell in the blood, and can inhibit the adhesion and activation of serum proteins and platelets[49-53]. Such surface coating materials have been produced by Biocompatibles International plc (Farnham, Surrey, UK). Albumin surface coating is also good and efficious anticoagulant coating. Albumin surface can effectively inhibit acute adhesion of fibrinogen both in vivo and in vitro, thus blocking platelet aggregation and fibrin formation. Another study demonstrated that the albumin-modified polyurethane surface are good blood compatible. The plasminogen activator-contained surface coating can cleavage the thrombus in vivo, because the plasminogen activator has fibrinolytic function. The plasminogen activators include fibrin plasminogen, polylysine, streptokinase and urokinase."

| [1] Boretos JW, Eden M, Contemporary Biomaterials: Material and Host Response, Clinical Applications, New Technology, and Legal Aspects. Park Ridge, NJ: Noyes Publications. 1984:232-233.
[2] March J. Advanced Organic Chemistry, Reactions, Mechanisms and Structure. 3rd ed. New York: John Wiley & Sons inc. 1985:216.
[3] Wang DA, Chen BL, Ji J, et al. Selective adsorption of serum albumin on biomedical polyurethanes modified by a poly(ethylene oxide) coupling-polymer with cibacron blue (F3G-A) endgroups. Bioconjug Chem. 2002;13(4): 792-803.
[4] Anderson AB, Tran TH, Hamilton MJ, et al. Platelet deposition and fibrinogen binding on surfaces coated with heparin or friction-reducing polymers. AJNR Am J Neuroradiol. 1996;17(5):859-863.
[5] Chen BL, Wang DA. Phase II study on surface construction and biocompatibility of polymer materials as cardiovascular devices: surface construction and biological responses. Zhongguo Zuzhi Gongcheng Yanjiu. 2014;18(21): 3412-3419.
[6] Chen BL, Wang DA. Surface construction and biocompatibility of polymeric used for cardiovascular medical device. Zhongguo Zuzhi Gongcheng Yanjiu. 2013;17(34):6183-6192.
[7] Chen BL, Wang DA. Hemocompatibility of biomedical polymeric materials: design of anticoagulant materials. Zhongguo Zuzhi Gongcheng Yanjiu. 2012;16(34): 6393-6396.
[8] Chen BL, Wang DA. Preparation and mechanism of anticoagulatent biomedical polymer materials with blood compatibility. Biocompatibility of a magnesium-zinc alloy implanted in rat cecum. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2011;15(29):5507-5510.
[9] Chen BL, Wang DA, Feng LX. Investigation on methods of surface modification of tissue engineering materials: polymer surface group transformation and bioactive molecule immobilization. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2010;14(3):552-554.
[10] Chen BL, Wang DA, Feng LX. Surface modification of tissue-engineered materials plasma and grafting modification. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2009;13(3):587-590.
[11] Chen BL, Wang DA, Feng LX. Application of polymer biomaterials in the tissue engineering. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2008;12(6): 1189-1192.
[12] Chen BL, Wang DA, Feng LX. Polymer porous membrane prepared using thermally induced phase separation. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2007;11(40):8217-8219.
[13] Chen BL, Wang DA, Feng LX. Topology of tissue engineering material surface for cell compatibility. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2007;11(18):3653-3656.
[14] Chen BL, Wang DA, Feng LX. Effects of physical and chemical properties of tissue engineered material surface on cell compatibility. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu. 2007;11(1):197-200.
[15] Chen BL, Wang DA, Feng LX. Cytological effect of tissue engineering materials with cell compatibility. Zhongguo Lincuang Kangfu. 2006;10(45):225-227.
[16] Chen BL, Wang DA, Feng LX, et al. The application of biomedical tissue engineering and the polymer tissue engineering material. Gaoshi Like Xuekan. 2007;27(1): 24-26.
[17] Chen BL, Wang DA, Feng LX, et al. Study on the blood compatibility of biomedical ploymer materials--project of antithromboeicity materials. Suihua Xueyuan Xuebao. 2007;27(1):186-188.
[18] Chen BL, Wang DA, Feng LX. Study on surfaces modify of the tissue engineering materials and application in the tissue engineering. Hulunbeier Xueyuan Xuebao. 2007; 15(1):52-54.
[19] Chen BL, Wang DA, Feng LX. Study on the tissue compatibility of biomedical ploymer materials--project of tissue-compatibility materials. Hulunbeier Xueyuan Xuebao. 2006;14(6):34-36.
[20] March J. Advanced Organic Chemistry, Reactions, Mechanisms and Structure. 3rd ed. New York: John Wiley & Sons Inc. 1985:1078.
[21] Brown EJ. Complement receptors, adhesion, and phagocytosis. Infect Agents Dis. 1992;1(2):63-70.
[22] Abele JE. Technical considerations: physical properties of balloon catheters, inflation devices and pressure measurement devices. In: Castaned-Zuniga WR, ed. Transluminal Angioplasty. New York: Thieme-Stratton, 1983:20-27.
[23] Triolo PM, Andrade JD. Surface modification and evaluation of some commonly used catheter materials. I. Surface properties. J Biomed Mater Res. 1983;17(1):129-147.
[24] Triolo PM, Andrade JD. Surface modification and characterization of some commonly used catheter materials. II. Friction characterization. J Biomed Mater Res. 1983;17(1):149-165.
[25] Salzman EW, Merill EW. Interaction of blood with artificial surfaces. In: Hemostasis and Thrombosis. 2nd ed. Philadelphia, PA, USA: J.B. Lippincott. 1987:1335-1347.
[26] Chenoweth DE. Complement activation in extracorporeal circuits. Ann N Y Acad Sci. 1987;516:306-313.
[27] Harrison JA, Colton RJ, White CT, et al. Atomistic simulation of the nanoindentation of diamond and graphite surfaces. Materials Research Sjociety, Symp. Proceeding. 1992;239:573.
[28] Hubbell JA, Massia SP, Desai NP, et al. Endothelial cell-selective materials for tissue engineering in the vascular graft via a new receptor. Biotechnology (N Y). 1991;9(6):568-572.
[29] Remes A, Williams DF. Immune response in biocompatibility. Biomaterials. 1992;13(11):731-743.
[30] Esmon CT. Thrombomodulin as a model of molecular mechanisms that modulate protease specificity and function at the vessel surface. FASEB J. 1995;9(10): 946-955.
[31] Plow EF, Herren T, Redlitz A, et al. The cell biology of the plasminogen system. FASEB J. 1995;9(10):939-945.
[32] Hårdhammar PA, van Beusekom HM, Emanuelsson HU, et al. Reduction in thrombotic events with heparin-coated Palmaz-Schatz stents in normal porcine coronary arteries. Circulation. 1996;93(3):423-430.
[33] Pasic M, Müller-Glauser W, von Segesser LK, et al. Superior late patency of small-diameter Dacron grafts seeded with omental microvascular cells: an experimental study. Ann Thorac Surg. 1994;58(3):677-684.
[34] Greisler HP, Klosak J, Dennis JW, et al. Endothelial cell growth factor attachment to biomaterials. ASAIO Trans. 1986;32(1):346-349.
[35] Gray JL, Kang SS, Zenni GC, et al. FGF-1 affixation stimulates ePTFE endothelialization without intimal hyperplasia. J Surg Res. 1994;57(5):596-612.
[36] Reilly CF, Kindy MS, Brown KE, et al. Heparin prevents vascular smooth muscle cell progression through the G1 phase of the cell cycle. J Biol Chem. 1989;264(12): 6990-6995.
[37] Au YP, Kenagy RD, Clowes MM, et al. Mechanisms of inhibition by heparin of vascular smooth muscle cell proliferation and migration. Haemostasis. 1993;23 Suppl 1:177-182.
[38] Kang SS, Gosselin C, Ren D, et al. Selective stimulation of endothelial cell proliferation with inhibition of smooth muscle cell proliferation by fibroblast growth factor-1 plus heparin delivered from fibrin glue suspensions. Surgery. 1995;118(2):280-287.
[39] Gosselin C, Ren D, Ellinger J, et al. In vivo platelet deposition on polytetrafluoroethylene coated with fibrin glue containing fibroblast growth factor 1 and heparin in a canine model. Am J Surg. 1995;170(2):126-130.
[40] Greisler HP, Ellinger J, Schwarcz TH, et al. Arterial regeneration over polydioxanone prostheses in the rabbit. Arch Surg. 1987;122(6):715-721.
[41] Greisler HP, Schwarcz TH, Ellinger J, et al. Dacron inhibition of arterial regenerative activities. J Vasc Surg. 1986;3(5):747-756.
[42] Greisler HP, Dennis JW, Endean ED, et al. Derivation of neointima in vascular grafts. Circulation. 1988;78(3 Pt 2):I6-12.
[43] Serruys PW, Emanuelsson H, van der Giessen W, et al. Heparin-coated Palmaz-Schatz stents in human coronary arteries. Early outcome of the Benestent-II Pilot Study. Circulation. 1996;93(3):412-422.
[44] Anderson AB, Enrico LS, Melchior MJ, et al., Photochemical immobilization of heparin to reduce thrombogenesis. Trans Soc Biomat. 1994;17(1):75.
[45] Clapper DL, Hagen KM, Hupfer JM, et al. Covalently immobilized ECM proteins improve patency and endothelialization of 4 mm grafts implanted in dogs. Trans Soc Biomat. 1993;16(1):42.
[46] Sauvage LR, Berger KE, Wood SJ, et al. Interspecies healing of porous arterial prostheses: observations, 1960 to 1974. Arch Surg. 1974;109(5):698-705.
[47] Williams SK, Schneider T, Kapelan B, et al. Formation of a functional endothelium on vascular grafts. J Electron Microsc Tech. 1991;19(4):439-451.
[48] Tweeden KS, Blevitt J, Harasaki H, et al. RGD Modification of Cardiovascular Prosthetic Materials, in Proceedings of the Cardiovascular Science and Technology Conference, Arlington, VA, Association for the Advancement of Medical Instrumentation. 1993;12:124.
[49] Campbell EJ, Chronos NA, Robinson KA, et al. Non-thrombogenic phosphorylcholine coatings stainless steel. Trans Soc Biomat. 1995;18(1):15.
[50] Chronos NA, Robinson KA, Kelly AB, et al. Thromboresistant phosphorylcholine coating for coronary stents. Circulation. 1995;92(suppl I):685.
[51] Ishihara K, Aragaki R, Ueda T, et al. Reduced thrombogenicity of polymers having phospholipid polar groups. J Biomed Mater Res. 1990;24(8):1069-1077.
[52] Eberhart RC, Munro MS, Frautschi JR, et al. Influence of endogenous albumin binding on blood-material interactions. Ann N Y Acad Sci. 1987;516:78-95.
[53] Grasel TG, Pierce JA, Cooper SL. Effects of alkyl grafting on surface properties and blood compatibility of polyurethane block copolymers. J Biomed Mater Res. 1987;21(7):815-842. |
| [1] | Chen Jinping, Li Kui, Chen Qian, Guo Haoran, Zhang Yingbo, Wei Peng. Meta-analysis of the efficacy and safety of tranexamic acid in open spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1458-1464. |
| [2] | Xu Jianxia, Wang Zhaoxu, Wang Chunren. Blood compatibility of disposable blood perfusion device in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 588-592. |
| [3] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [4] | Lang Limin, He Sheng, Jiang Zengyu, Hu Yiyi, Zhang Zhixing, Liang Minqian. Application progress of conductive composite materials in the field of tissue engineering treatment of myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3584-3590. |
| [5] | Jing Jinpeng, Zhang Yue, Liu Xiaomin, Liu Yi. Buyang Huanwu Decoction in prevention of deep venous thrombosis after orthopedic surgery: meta-analysis and trial sequential analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3267-3274. |
| [6] | Zhou Qi, Gao Yi, Wei Kang, Li Jun, Xu Jianda, Jiang Yang, Qu Yuxing. Total knee arthroplasty for rheumatoid arthritis: knee function and biochemical index changes [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(9): 1337-1341. |
| [7] | Wei Yong, Li Jun, Zhang Yong, Yu Hao, Xie Jia. Prevalence rate and high-risk factors of preoperative lower-limb deep venous thrombosis in patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(27): 4338-4342. |
| [8] | Gao Zhaopeng, Gao Ming, Jiang Zhen, Zhang Qinming, Jia Dailiang, Wang Haibin. Hemostatic and anticoagulant effects of proximal femoral nail anti-rotation in repairing elderly patients with intertrochanteric fractures during perioperative period [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(21): 3315-3320. |
| [9] | Yu Dongming, Hu Song, Yang Xiaozhong, Zou Yi, Chen Yong, Hu Bo, Gao Daxin. Tranexamic acid controls postoperative hidden blood loss in senile intertrochanteric fracture [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(21): 3353-3357. |
| [10] | Luo Zhihong, Yang Fan, Yang Peng, Chen Xiaojun, Gong Shuidi, Pang Fengxiang, He Xiaoming, Shen Yingshan, Li Weifeng, Chen Lixin, Wei Qiushi, He Wei, Chen Zhenqiu. Effect of platelet-rich plasma on blood coagulation indexes after hip preserving surgery for osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(15): 2393-2398. |
| [11] | Yuan Bo, Wang Zhiwei, Tang Yifan, Zhou Shengyuan, Chen Xiongsheng, Jia Lianshun. Construction of polycaprolactone-tricalcium phosphate with different mixture ratios using three-dimensional printing technology and its osteoinductivity in vitro [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(6): 821-826. |
| [12] | Wang Yang, Nie Jinshan, Gu Zhun, Zhu Kai. Construction and in vitro evaluation of a biodegradable cationic gene delivery system based on hyperbranched polyamidoamine [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(6): 936-944. |
| [13] | Leng Yi, Li Zuhao, Ren Guangkai, Wang Zhonghan, Gao Chaohua, Shi Chenyu, Liu He, Wu Dankai. Application and progress of bioactive scaffolds in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(6): 963-970. |
| [14] | Wu Xingyuan, Zhang Guoru, Liu Tang, Zhou Caisheng. Intravenous and intraarticular tranexamic acid can reduce blood loss and inflammatory response during cemented posterior cruciate ligament-retaining unilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(36): 5753-5759. |
| [15] | Han Guangtao, Li Haohuan. Influence of the concept of fast track surgery on the physiological and psychological rehabilitation of patients undergoing total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(36): 5760-5765. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||